 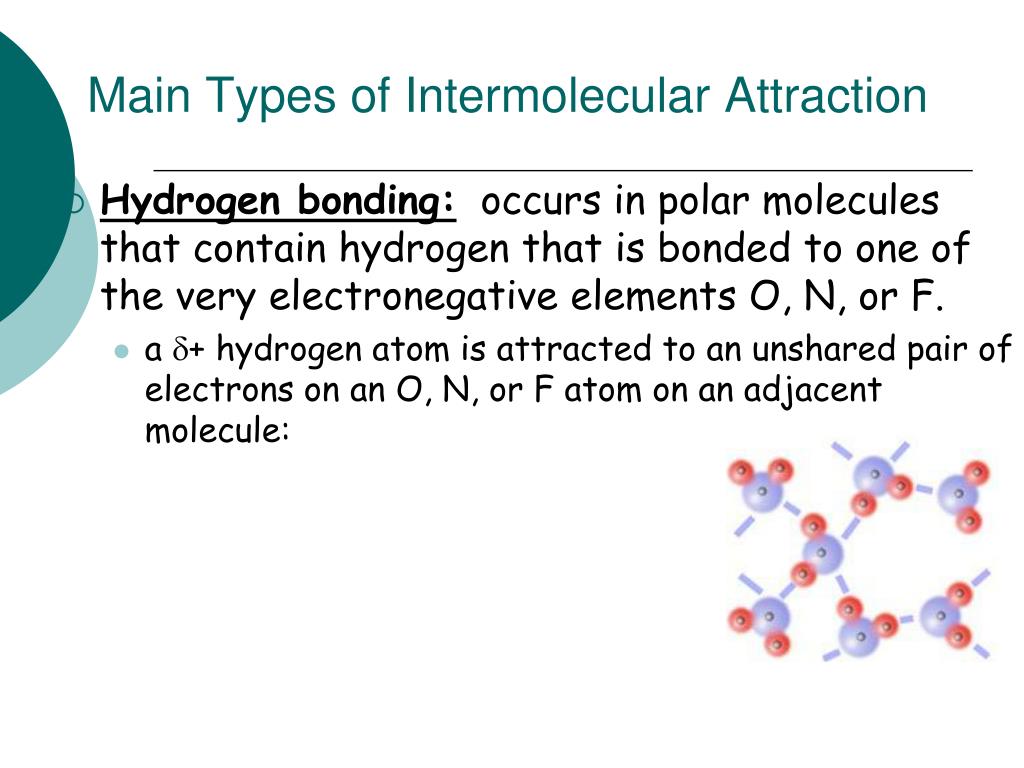
Solution: Helium gas, Acetone, Water, Isobutyl alcoholĮxplanation: Since having stronger intermolecular forces increases boiling point, this problem is to rank the compounds from weakest to strongest intermolecular forces. Influence of Intermolecular Forces Solutions Order the following compounds from lowest boiling point to highest: Influence of Intermolecular Forces Practice Problems Polar molecules and nonpolar molecules don’t dissolve well with each other. Polar molecules dissolve polar molecules through dipole-dipole forces, and nonpolar molecules dissolve nonpolar molecules through London dispersion forces. The phrase “like dissolves like” helps predict the solubility of a solute in a solvent. The stronger the intermolecular forces between the solute and solvent, the greater the solubility. Source.Īnother one of the effects of intermolecular forces is solubility. A water droplet maintains its “bubble” shape because of surface tension. The stronger the intermolecular forces, the greater the surface tension. This property is caused by intermolecular forces, which keep the molecules together and cause the surface of the liquid to resist external forces. Surface tension is the elastic property of the surface of a liquid, such as how water drops form a spherical shape. One of the effects of intermolecular forces is surface tension. This makes it harder for the liquid to evaporate, resulting in lower volatility and lower vapor pressure. Stronger intermolecular forces give a liquid a stronger attraction to itself. An increase in volatility, which is the tendency of a substance to evaporate, results in an increase in vapor pressure. Vapor pressure is the pressure exerted by vapor on the surface of a liquid. The strength of the intermolecular forces of a liquid affects the liquid’s vapor pressure. Honey is sticky and flows slowly due to its strong intermolecular forces. This is why water flows very well while glycerol doesn’t. Glycerol, for example, has much stronger intermolecular forces than water because it contains 3 -OH hydroxyl groups capable of hydrogen bonding, while water only has one. Stronger intermolecular forces cause a fluid to have a stronger attraction to itself, giving it a high viscosity. Viscosity is a result of intermolecular forces. A high viscosity results in a thicker and stickier fluid. Viscosity is the measure of a fluid’s resistance to flow. ViscosityĪnother effect of intermolecular forces is viscosity. Propane has such a higher boiling point than methane because it has stronger London dispersion forces (larger molecule) and stronger IMFs overall than methane. Therefore, if a substance has stronger IMFs, it is harder to pull its molecules away from each other, resulting in the substance having a higher boiling point and melting point.įor example, propane (C3H8), has a boiling point of -42☌ while methane (CH4), has a boiling point of -161.6☌. Having strong intermolecular forces means that the attraction between the molecules is strong. The boiling point is the temperature at which a liquid can turn into a gas, and the melting point is the temperature at which a solid can turn into a liquid. How Intermolecular Forces Influence Physical Properties Boiling/Melting points Intermolecular forces have varying strengths, and the strength of the intermolecular forces within a substance influences the substance’s physical properties, including boiling/melting points, viscosity, surface tension, vapor pressure, etc. There are different types of intermolecular forces: London dispersion forces, dipole-dipole forces, ion-dipole forces, and hydrogen bonding. 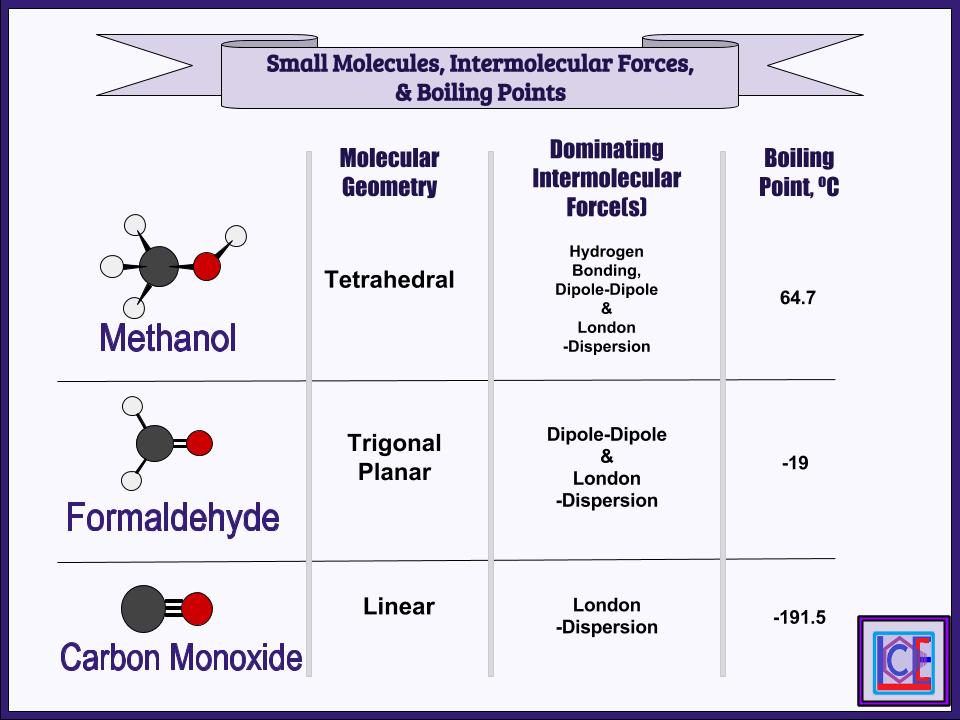
Though IMFs include both attractive and repulsive interactions, the term is usually only used for attractive interactions between molecules (having strong IMFs between two molecules means the force of attraction between the two is strong). They are electrostatic interactions between charged molecules. 
Intermolecular forces (IMFs), as its name would suggest, are forces between molecules. Properties of Solids, Liquids, and Gases.You will understand how intermolecular forces influence the physical properties of matter, such as melting point, viscosity, and surface tension. In this article, you will learn about the effects of intermolecular forces (IMFs). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
